Policy & Regulation
French Agency Issues Warning on Glyoxylic Acid in Hair Straightening

French Agency Warns Glyoxylic Acid: Risks to Health and Cosmetics
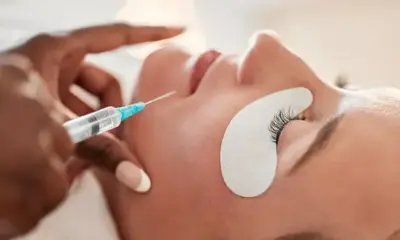
The French Agency Warns Glyoxylic Acid due to its confirmed risk of causing acute renal failure. On January 23, French health authorities reiterated their recommendation not to use hair straightening products containing glyoxylic acid. This warning comes after reports of severe health risks linked to the substance, prompting authorities to urge further investigations and regulatory actions.
French Agency Warns Glyoxylic Acid: Health Risks and Recommendations
In mid-October, the French Agency Warns Glyoxylic Acid, advising both hair salons and individuals to avoid using hair straightening products containing this substance. The French Agency for Food, Environmental, and Occupational Health Safety (ANSES) highlighted the importance of precautionary measures, as they awaited the conclusions of an expert report. The risk was initially identified due to severe adverse reactions experienced by women exposed to these products during hair straightening procedures.
The Agency now considers it “highly probable” that glyoxylic acid is responsible for causing acute renal failure in individuals. When absorbed through the scalp, it transforms into calcium oxalate crystals that damage the kidneys. This has led ANSES to call for a European-level risk assessment, urging the European Union to consider limiting or even banning the substance in cosmetics.
Health Concerns Linked to Glyoxylic Acid in Hair Products
The French Agency Warns Glyoxylic Acid for another crucial reason. Glyoxylic acid can break down into other harmful substances, such as formaldehyde, during the hair-heating phase. Formaldehyde is a carcinogen, making its release in hair straightening treatments even more concerning. ANSES urges that other products potentially breaking down into glyoxylic acid should be identified to prevent further risks.
In Israel, glyoxylic acid-containing hair straightening products have already been banned since 2022 due to similar health risks, including 26 reported cases of acute renal failure. These incidents led to symptoms such as abdominal pain, nausea, and vomiting. The kidney functions of the affected individuals returned to normal after rehydration treatments.
Call to Action for Safer Cosmetic Practices
ANSES encourages all marketers to review the safety of products containing glyoxylic acid. It also urges the need for clear precautions to ensure safe usage. As the regulatory discussion continues at the European level, it is essential to stay informed about the evolving stance on glyoxylic acid’s use in cosmetics.
For more updates on health and safety in cosmetics, explore more news on this website.